|
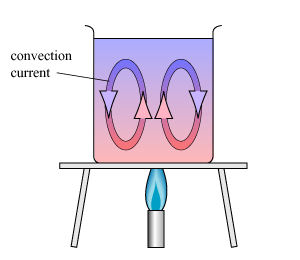
Radiation is the last but definitely not least of the ways in which heat transfers and moves from one place to another. The Earth's largest contributor of energy is the SUN! The sun sends out heat energy in the form of radiant energy waves. The two main energy waves are known as Ultraviolet A and Ultraviolet B. Radiation does NOT require particles to move through the atmosphere (Thank goodness for that or we'd be popsicles!). Instead heat just moves in waves! Much like how sound and light travels in waves. These radiant energy waves are what makes us feel warm but it is also what causes us to burn if we are out under the sun for too long without protection. Certain types of surfaces will absorb or reflect radiation waves. Record down some examples of different surfaces that 1) absorb heat radiation more than others, and 2) reflect radiation more than others. Copy a diagram of the radiometer and explain how it works. Eureka and Radiation: Have you ever heard the expression that "Warm or Hot air rises"? Well, Convection explains this phenomenon. Convection is the way heat energy travels in liquids and gases using particles. Due to the increased movement of particles and therefore spreading out of the particles, heated areas of liquids and gases are much lighter than areas that have less heat. The particles in the warmer area are less dense (more spread out) than particles in a cooler area which are closer together. This means that warmer air is lighter, and that is why warm/hot air rises!
Here is another Eureka 5 minutes! SOME CONVECTION OCCURRING IN REAL LIFE! Directly below is how Convection currents shape our planet and its landforms. The Mantle is full of Magma (molten rock) that heats from the core of the Earth. Because the core is the heat source, the warmer magma pushes upwards and cooler magma near the surface of the planet, sinks downwards. This constant but very slow movement is behind Plate Tectonics. Watch below!! In this video, you will see how our weather is a result of how heat moves on our planet. From the Sun's radiation, to Conduction of heat on surfaces on the planet, to convection currents in the air and in the water... all these methods of heat transfer act to move and shift the winds and water, creating all the weather patterns we see and feel around the globe! Cool!! Conduction is one way that Heat Energy moves from one place to another. In conduction particles in solids are heated. This heat energy causes the kinetic energy to increase in particles making particles bump into each other, vibrating more, needing more space. As particles bump into each other, bumping into particles that are not being heated directly, the heat energy starts to spread and move through the solid. This is known as Conduction. Some materials are better conductors than others. If a material is not a good conductor at all, we call that an insulator! In class you will be designing your own experiment to test the conductivity of a variety of materials including: aluminum, copper, brass, wood, glass, and plastic. You will have a choice of various shapes of these tubes ranging from thin, hollow, and thick. Think of a QUESTION or PURPOSE for your experiment. Examples might include: - Does the amount of matter of a specific material make a difference in how fast a material conducts heat? - Which material conducts heat the fastest out of wood, glass, metal? - Which metal conducts heat the fastest? Aluminum, copper, or brass? Once you have identified the purpose of your experiment, write a hypothesis (prediction) and explain your thinking based on what you know already. Circle the materials you will need. Hand it in to Ms. Ang for approval and you will be given materials to start conducting your experiment! Have fun! Be safe!! If heat causes particles to move around more, spread apart, and take up more space, then an object that is heated must expand (get bigger) and objects being cooled must contract (get smaller)! The BALL and RING Demo: Make a prediction what will happen during the demonstration. Record this on lined paper. Watch the demonstration. Record what happens in 3 or 4 steps. Explain what happened and why using the Particle Theory. Level 3 responses will include: Particle Theory vocabulary, as well as the word, "expand" and "contract" Level 4 responses will include the above, plus a real life example of how matter expands and contracts. To be collected next class. Here's a Eureka Video showing Expansion and Contraction of matter and why it occurs using the Particle Theory: The Particle Theory of Matter is a theory that scientists have used for decades to help explain how and why matter reacts the way it does to temperature and to other matter; however, it is important to note that it IS a THEORY. The definition of a THEORY is as follows: "When used in non-scientific context, the word “theory” implies that something is unproven or speculative. As used in science, however, a theory is an explanation or model based on observation, experimentation, and reasoning, especially one that has been tested and confirmed as a general principle helping to explain and predict natural phenomena."1 So, based on decades of observation and experimentation, the Particle Theory is a valid context for understanding matter and its behaviour. The problem is, we can't actually 'see' the particles moving and vibrating, we just simply theorize that they are there, acting in a certain way because it makes sense. Note that the word, "Particle" can refer to a molecule, atom, or even an ion (smaller part of an atom). Basic principles behind the Particle Theory are: 1) All matter is made up of extremely tiny particles. 2) These tiny particles move and vibrate continuously carrying kinetic energy (energy of motion) 3) Adding heat to these particles cause an increase in energy which causes the particles' movement to increase This means: the more heat, the more the particles move. 4) Particles have different levels of attraction with each other and stay together with chemical bonds (Like how + and - magnets may attract) 5) When particles move more because of heat, they need more space and their bonds weaken. Particles spread apart. 6) Particles of different pure substances are different from one another! Watch the video below. The first one is the most interesting and gives you a good overview of the principles behind the Particle Theory of Matter. The second one is a dramatization. I found my favorite series of Science videos, "Eureka". They are animated, short, and descriptive and of course, they are on Youtube! Watch this series in order here....
Temperature: Is the measure of how much Heat Energy is present. The temperature of something depends upon how fast its tiny particles of matter are moving. Matter that has faster moving particles have a higher temperature than matter with slower moving particles.
Heat: Is the energy that is transferred (moved) from matter at a higher temperature to matter at a lower temperature. It is also called THERMAL energy. |
Archives
March 2015
Categories |
Heat and Temperature
Proudly powered by Weebly

 RSS Feed
RSS Feed