Matter is anything that takes up space and has mass. Basically, it is everything, and anything!
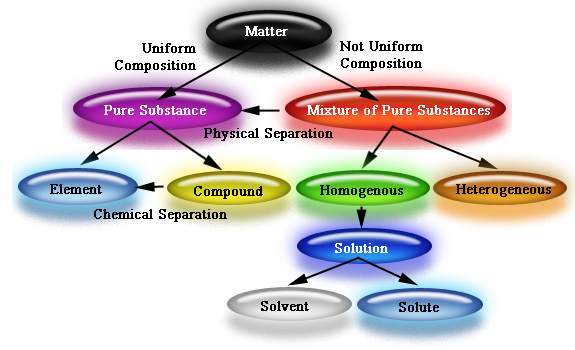
We are used to classifying matter into solids, liquids, and gasses as we have been doing this since kindergarten! This year, we will learn to understand matter in a different way, categorizing matter by its appearance and composition. The following chart gives an overview.
We are used to classifying matter into solids, liquids, and gasses as we have been doing this since kindergarten! This year, we will learn to understand matter in a different way, categorizing matter by its appearance and composition. The following chart gives an overview.
Matter can be classified as a Pure Substance or a Mixture; Homogenous or Heterogeneous.
Homogeneous: no visible different parts, appears the same throughout.
Heterogeneous: different parts are visible, does not look the same throughout.
Pure substances: homogenous in appearance and may be either a pure element such as Gold, or a compound of elements, such as water H20 (Hydrogen and Oxygen together)
Mixtures: can be either Homogeneous or heterogeneous and are made up of a combination of different types of matter.
Heterogeneous Mixtures: Mechanical Mixtures such as a bowl of cereal with milk and bananas
Homogeneous Mixtures: Mixtures that involve a Solute and Solvent to make a Solution
Below is a video that helps reinforce the concept of Homogeneous and Heterogeneous and also refers to the THREE COMMON BEVERAGES lab that we did in class.
Homogeneous: no visible different parts, appears the same throughout.
Heterogeneous: different parts are visible, does not look the same throughout.
Pure substances: homogenous in appearance and may be either a pure element such as Gold, or a compound of elements, such as water H20 (Hydrogen and Oxygen together)
Mixtures: can be either Homogeneous or heterogeneous and are made up of a combination of different types of matter.
Heterogeneous Mixtures: Mechanical Mixtures such as a bowl of cereal with milk and bananas
Homogeneous Mixtures: Mixtures that involve a Solute and Solvent to make a Solution
Below is a video that helps reinforce the concept of Homogeneous and Heterogeneous and also refers to the THREE COMMON BEVERAGES lab that we did in class.
BIG IDEA: What would you have to do in order to determine if a substance was homogeneous or heterogeneous???
 RSS Feed
RSS Feed